How FDA's top vaccines official is timing his COVID and flu shots
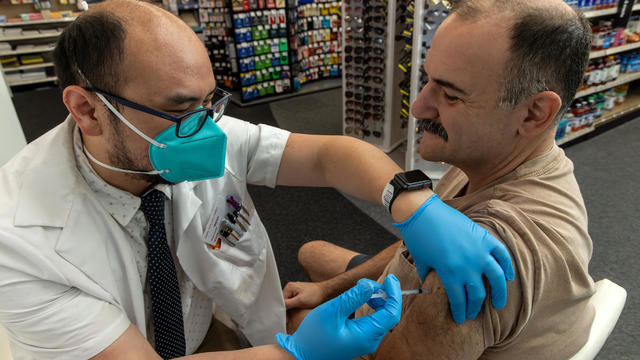
Can you get your COVID booster and flu shot at the same time? Here's what health experts say.
Watch CBS News
Alexander Tin is a digital reporter for CBS News based in the Washington, D.C. bureau. He covers federal public health agencies, including the response to infectious disease outbreaks like COVID-19. Previously, he was a campaign reporter for CBS News based out of Las Vegas, where he was raised. He covered presidential, Senate and House candidates for the 2020 election cycle in Arizona, California, Nevada and New Mexico. He has also worked in Washington for "Face the Nation" and in New York for the "CBS Evening News." Tin graduated from Columbia University in 2017 with a bachelor's degree in political science.

Can you get your COVID booster and flu shot at the same time? Here's what health experts say.
The Biden administration is also pouring $600 million into more COVID tests manufacturing across the U.S.

ASR Pharma said it was surprised and "deeply disappointed" by the FDA request for more trial data on its Neffy nasal spray for severe allergic reactions.

The variant, which has an unusually large number of mutations, has also been spotted in numerous countries, but remains rare for now.
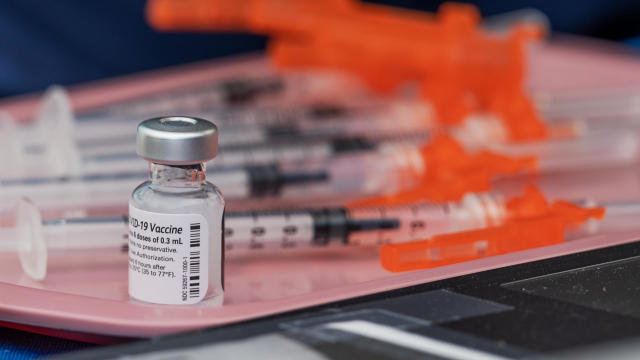
The new COVID-19 vaccines for fall 2023 started rolling out this week.
Vaccine experts considered narrower recommendations for only higher-risk groups, but decided almost all Americans could benefit.
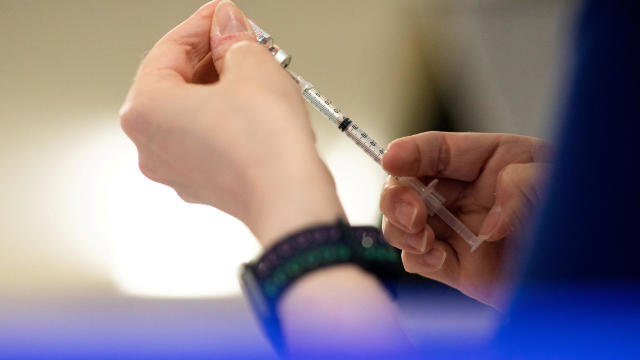
The new shots are similar to previously approved formulas but were updated to target strains of the virus descended from Omicron.

New COVID vaccines are expected after Sept. 12.

Rates have nearly doubled over the past week.

Officials say testing should still work for the new COVID variant.

So far, the CDC doesn't think the highly mutated BA.2.86 variant is to blame.

Expectant parents could soon have another new option this fall to protect their newborns from RSV, the most common cause of hospitalization in American infants.

Expectant parents could soon have another new option this fall to protect their newborns from RSV, the most common cause of hospitalization in American infants.

On social media, the variants have been nicknamed "Eris," "Fornax" and "Pirola."
More pressing, hospitalizations have risen by nearly 27%. That marks the fourth-largest growth in the country currently among states.