COVID Vaccine Booster Shots Rollout To Begin September 20, Starting With Those Vaccinated First
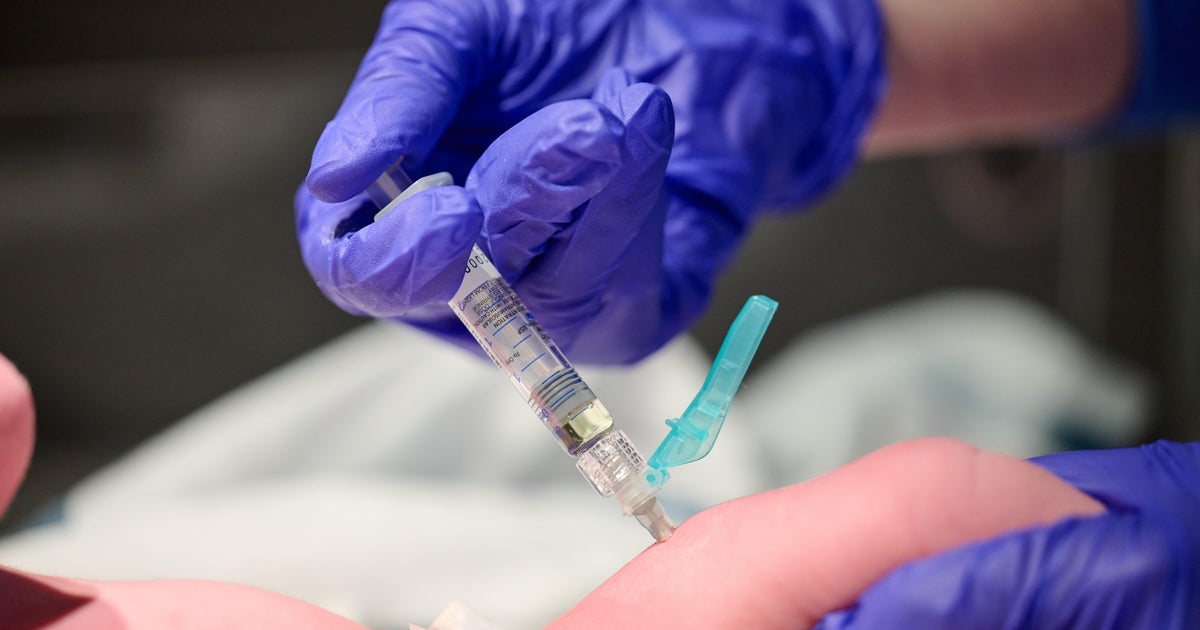
(CNN) -- US health officials and medical experts announced in a joint statement on Wednesday that booster doses of Covid-19 vaccine will be offered this fall, subject to authorization from the US Food and Drug Administration and sign off from the US Centers for Disease Control and Prevention.
"We are prepared to offer booster shots for all Americans beginning the week of September 20 and starting 8 months after an individual's second dose," US health officials, including CDC Director Dr. Rochelle Walensky and FDA Acting Commissioner Dr. Janet Woodcock, said in the statement.
"At that time, the individuals who were fully vaccinated earliest in the vaccination rollout, including many health care providers, nursing home residents, and other seniors, will likely be eligible for a booster. We would also begin efforts to deliver booster shots directly to residents of long-term care facilities at that time, given the distribution of vaccines to this population early in the vaccine rollout and the continued increased risk that COVID-19 poses to them," the statement said.
The officials write that the authorized Covid-19 vaccines are "remarkably effective in reducing risk of severe disease, hospitalization, and death, even against the widely circulating Delta variant," but it's clear that protection against the coronavirus begins to decrease over time.
The statement said "current protection against severe disease, hospitalization, and death could diminish in the months ahead, especially among those who are at higher risk or were vaccinated during the earlier phases of the vaccination rollout. For that reason, we conclude that a booster shot will be needed to maximize vaccine-induced protection and prolong its durability."
Officials are expected to discuss the plan during at 11 a.m. ET White House Covid-19 briefing. The statement was signed by Walensky, Woodcock, Surgeon General Dr. Vivek Murthy, National Institutes of Health Director Dr. Francis Collins, National Institute of Allergy and Infectious Diseases Director Dr. Anthony Fauci, Assistant Secretary for Health Dr. Rachel Levine, Chief Science Officer for the COVID Response Dr. David Kessler and Chair of the COVID Health Equity Task Force Dr. Marcella Nunez-Smith.
While those initial booster doses will be for the Pfizer/BioNTech and Moderna vaccines, the officials noted in the statement that they anticipate booster shots will likely be needed for people who initially received the Johnson & Johnson vaccine. "Administration of the J&J vaccine did not begin in the U.S. until March 2021, and we expect more data on J&J in the next few weeks," the statement said.
The officials ended the statement by noting that they continue to expand efforts to increase the supply of vaccines globally for other countries, "building further on the more than 600 million doses we have already committed to donate globally."
Push for more data
For months, federal health officials and agencies had maintained that boosters for the general public were not needed, but said they were watching the data closely.
Last week, the FDA authorized third doses for some people who are immunocompromised and the US Centers for Disease Control and Prevention almost immediately recommended giving those doses. But the agency also said bosoters were not yet necessary for the rest of the country.
At the White House Covid-19 briefing, officials are expected to present three sets of data that suggest waning vaccine protection against Covid-19 infection in the United States, a senior federal health official told CNN Chief Medical Correspondent Dr. Sanjay Gupta.
The three data sets were said to include 10 million vaccinated New Yorkers, data from long-term care facilities, and data from a group of 21 hospitals.
The official cautioned that while the data suggests decreased protection against infection, it's still believed there has not been a decline in protection against hospitalization and death from Covid-19 for now.
The official also cautioned that the FDA would still need to carefully review safety data to ensure there was no danger from "hyperimmune response" from a third dose. So far, only Pfizer has submitted such data for review.
Dr. Paul Offit, a vaccine expert and adviser to the FDA, told CNN's Erin Burnett on Tuesday that the US needs clear data from the CDC to support the need for Covid-19 vaccine boosters for the general public.
"It is frustrating," Offit said. "You would like to think that the data would come first and then the recommendation would come second, because right now, we're all guessing what's going on."
Offit noted that just days ago, the CDC presented data showing that Covid-19 vaccines still protect against severe disease. In a call with clinicians on Tuesday afternoon, CDC officials said the timing of a booster dose of coronavirus vaccine has not yet been determined.
"What I hope doesn't happen is that we are seeing the sort of fading in immunity against asymptomatic infection or mildly symptomatic infection, because that has to happen at some level," Offit said. "The goal is to protect against severe, critical disease. To date, it appears the vaccines were doing it."
Other experts have also cast doubt on whether booster shots are needed, saying it's far more important for more people to be fully vaccinated in the first place.
"If you think a third dose of the vaccine is going to end the pandemic, then you are kidding yourself," Scott Hensley, an immunologist at the University of Pennsylvania, told CNN. "The way to end this pandemic is to get the vaccine distributed across the globe."
The-CNN-Wire
™ & © 2021 Cable News Network, Inc., a WarnerMedia Company. All rights reserved.