
Children's COVID-19 Hospitalizations Rise In Maryland; Local Pediatrician Says Kids Are Increasingly Vulnerable
As of Tuesday, there are nineteen children in the hospital battling COVID in Maryland.
Watch CBS News

As of Tuesday, there are nineteen children in the hospital battling COVID in Maryland.
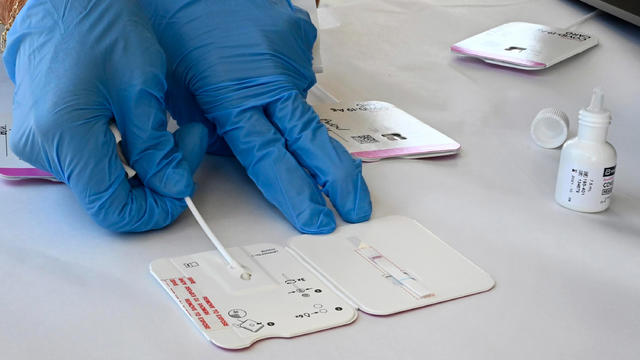
None of the counties have hinted at when a restock is in sight.

When students return to the University of Maryland College Park campus for classes between Jan. 3 and 21, they will have to show a negative COVID-19 test before their first in-person class and will have to wear KN95 masks in class amid a surge in cases, according to an email from President Darryll J. Pines and Jennifer King Rice, senior vice president and provost.

Hampden United Methodist Church held its second Christmas Eve service Friday night. This is one of many churches whose doors were closed for the holidays last year, but now because of safety protocols like masks and vaccinations, they feel like they now can come together as a church community.

All in-person programs and events at Baltimore County Public Libraries will be suspended, effective Monday, until further notice due to the COVID-19 surge, Baltimore County officials said Thursday.


A WebMD study found 57% of us are grieving someone we lost in the last three years.

With hospitalizations reaching 1,345 as of Tuesday in Maryland, strained hospital systems are concerned over upcoming holiday travel and the Omicron variant of COVID-19.
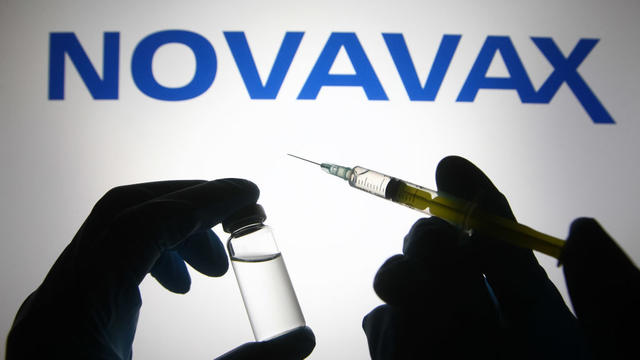
Novavax has been given emergency use authorization in Indonesia and the Philippines, has applications pending with the World Health Organization and Britain, and plans to file with the U.S. Food and Drug Administration by year's end.

As the virus spreads, more cities are adding restrictions, including New York and Washington, DC.

Sheppard Pratt held its grand opening Thursday for the Center for Autism, which will offer diagnostic services and subsequent case management for individuals throughout their lifespan.

Meal distributions next week will occur on Monday through Wednesday between 10 a.m. and noon, and information on January meals are forthcoming.

COVID-19 outbreaks are causing local universities to cancel events and change how the fall semesters conclude.
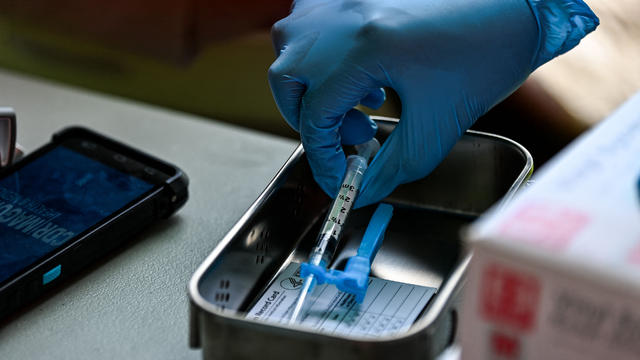
The new recommendation: "mRNA vaccines are preferred over the Janssen Covid-19 vaccine for the prevention of Covid-19 for those 18 years of age and over."

Historically, most students who receive such meals have applied for the program.

Almost two weeks after the heaviest snow and sleet storm in a decade, Maryland is bracing for the coldest temperatures this winter.

First Alert Weather Days continue Saturday overnight and through Sunday due to the risk of extreme cold.

A man was hospitalized following a shooting in Southeast Baltimore early Saturday morning, police said.

BGE announced layoffs affecting dozens of union members, according to a statement from the IEBW Local 410 on Friday.

A Baltimore County mom turned to WJZ after saying Meta wrongfully disabled her daughter's Instagram, where she shared her dog-training and performance content.

The Ravens said they "clearly made an error" and won't appeal the ruling.

A Howard County Police officer responded to a call last weekend that really got his goat.

Chilly night ahead with some frost possible west of I-95. Warmer weather returns this weekend.

Storm threat continues Thursday with high heat added to the mix too during the afternoon.
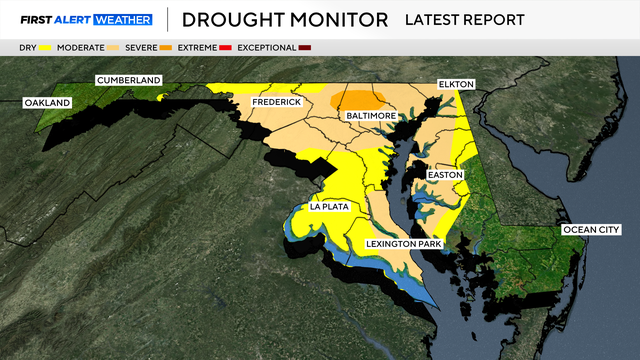
Drought conditions in Maryland have improved after several rounds of wet weather in May.

Coach Tre shares a few beginner-friendly workout moves for those who are looking for a fresh start. He focuses on nutrition, physical, and mental heath in all his clients.

Marty Bass is with Chef Antoine Thomas learning how he uses his special Chef's Blend seasoning to create the ultimate game day spread.

WJZ visited the special Valentine's Day edition of the haunted attraction. Cupid's Revenge, presented by Bennett's Curse, is open February 6th and 7th.

The Chesapeake Bay Boat Show returns to Maryland for its 5th year.

Maryland's biggest flag distributor, CRW Flags, gets ready for Fourth of July.

The Maryland No. 1 has symbolized dominance for decades. It's one of the most coveted jerseys in college lacrosse.

Buzz Williams' first season at Maryland is producing the wrong kind of history.

The Baltimore Orioles acquired utility player Blaze Alexander in a trade on Thursday with the Arizona Diamondbacks, who received reliever Kade Strowd and minor leaguers Wellington Aracena and José Mejia.

Doyle takes over a unit that was phenomenal in 2024 before regressing this season as two-time MVP quarterback Lamar Jackson dealt with injury problems.

Jesse Minter has the job, and with it the task of replacing John Harbaugh at the helm of a team that was considered a Super Bowl favorite just a few months ago.

Colin Demarco was arrested in January, months after he was seen in a Ring camera image at Vought's door, wearing a surgical mask and gloves.

Former Capitol Police officer Harry Dunn — who emerged as a national figure after the Jan. 6 riot — announced a second bid for Congress on Wednesday.

Five-year-old Liam Conejo Ramos and his father were released from ICE custody on Sunday, a day after a federal court ordered their release.

The Senate passed a deal on a package of spending bills late Friday, sending it to the House, though funding for dozens of government agencies lapsed until the House takes it up.

Mr. Trump previously threatened to impose tariffs against countries that do not support his plans to acquire the territory.

Nearly 200 families were expected to shop at the 10th annual Christmas Store, hosted by the University of Maryland, Baltimore, and The Foundry Church on Friday.

After a loved one became a victim of sexual assault, a Maryland family is on a mission to prevent it among teens.

Over 400 future star chefs took over Baltimore on Saturday for a National Culinary Cook-off.

Chopped Broadway Bodega & Deli is making its mark as the first Black- and women-owned bodega in Fells Point, bringing the unmistakable flavors of a classic New York bodega to Baltimore.

'I Believe in Me Girls' held its fifth annual workshop on Saturday to empower young women while introducing them to a variety of career opportunities in the creative industry.

Senator Kagan said this bill will limit what she calls "bait and switch" and price gouging.

The Maryland Department of Emergency Management increased the State Activation Level from Normal to Partial on Friday morning in response to a Microsoft outage that has paralyzed systems worldwide.

Neighbors in one west Baltimore neighborhood are figuring out what to do for groceries, due to the Giant Food location in Edmondson Village closing next month.

A string of recalls connected to Wisconsin-based supplier Schreiber Foods has now extended to Aldi stores after similar recalls at Hy-Vee and other grocery chains.

The U.S. Drug Enforcement Administration is asking to move marijuana to Schedule III, alongside ketamine and some anabolic steroids

New flights to both U.S. and Caribbean destinations are coming to BWI in 2026.

After three decades of serving customers on the Baltimore waterfront, The Cheesecake Factory is closing its Inner Harbor location.

The company voluntarily issued the recall on September 23.

The Baltimore Tech Hub, one of 31 federal "Tech Hubs" designated last year, was skipped for a cut of $504 million in the first round of funding for the program.

Primanti Bros. on Tuesday announced plans to expand to the Baltimore region this spring.

Mixed nuts from Ohio-based Mellace Family Brands sold at some Wegmans stores could be tainted with Salmonella, FDA warns.

The company voluntarily issued the recall on September 23.

With open enrollment starting soon, employees could get sticker shock, with their costs expected to rise 6% to 7% for 2026, a new study says.

After WJZ shed light on a rat infestation plaguing one Towson community, Baltimore County officials say they are now taking action.

Recalled supplemental shakes were distributed to hospitals and long-term care facilities nationwide, federal officials say.

Music's biggest night returned Sunday with the 68th annual Grammy Awards. Here is how to watch and stream and what to know.

James Ransone, the actor who played Ziggy Sobotka in the HBO series "The Wire" and appeared in many other TV shows and movies, has died.

Director Rob Reiner and wife, Michele, were found dead in their Los Angeles home, sources told CBS News, in what police called an apparent homicide.

President Trump is hosting the Kennedy Center Honors on Sunday after presenting medals to the 2025 Kennedy Center honorees during an Oval Office ceremony Saturday.

A Maryland ticket broker is accused of illegally purchasing and reselling hundreds of thousands of tickets for profit, including thousands to Taylor Swift's highly-sought Eras Tour.
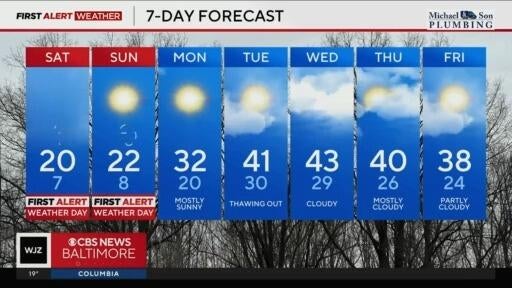
Strong wind gusts and frigid temperatures are expected in Maryland this weekend.
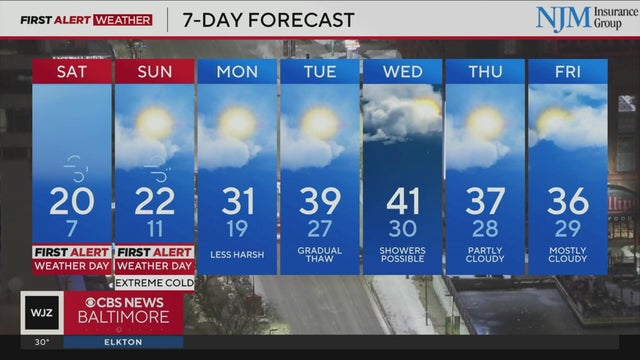
Cutter Martin has your Friday evening forecast | 2/6/2026
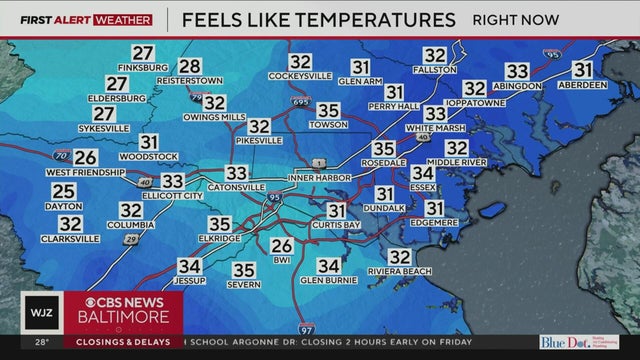
Parts of Maryland will see some light snowfall on Friday before frigid temperatures set in for the weekend.

Saturday and Sunday are First Alert Weather Days due to the risk of extreme cold.

WJZ-TV meteorologist Steve Sosna has your latest weather forecast.

Strong wind gusts and frigid temperatures are expected in Maryland this weekend.

Anne Arundel County police say a woman was taking out her trash in Glen Burnie Thursday night when a man sexually assaulted her and tried to kidnap her.

Students at several Maryland high schools participated in walkouts on Friday to make their voices heard and speak out against ICE operations locally and nationwide.

Almost two weeks after the heaviest snow and sleet storm in a decade, Maryland is bracing for the coldest temperatures this winter.

The Maryland No. 1 has symbolized dominance for decades. It's one of the most coveted jerseys in college lacrosse.